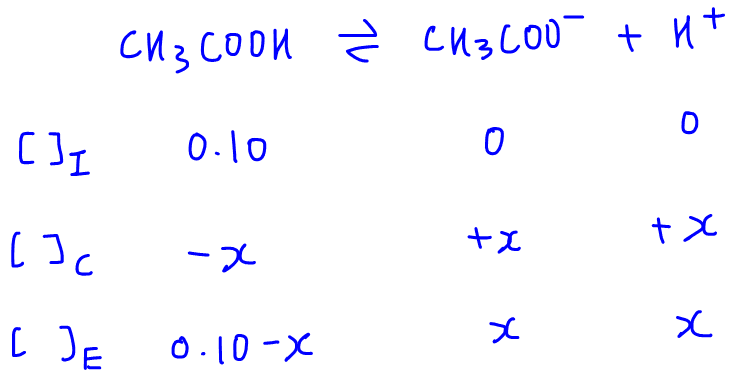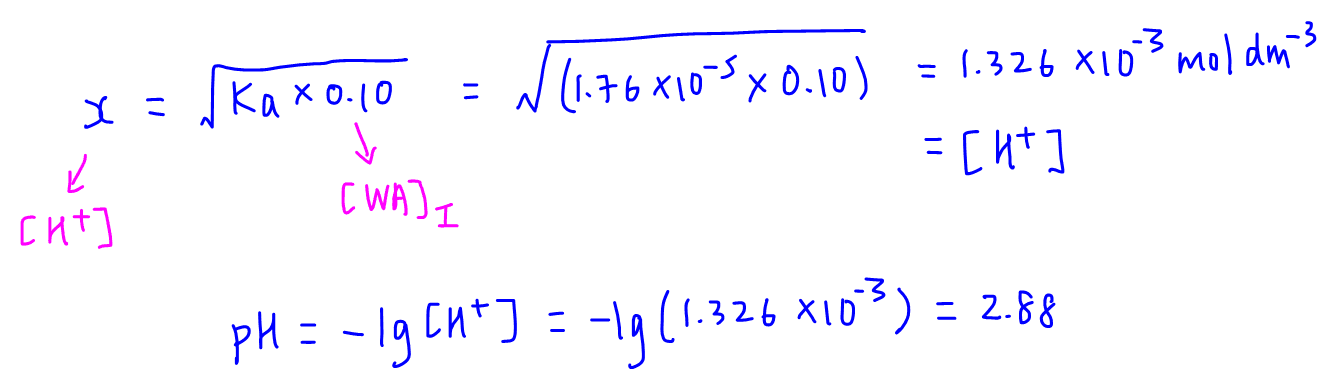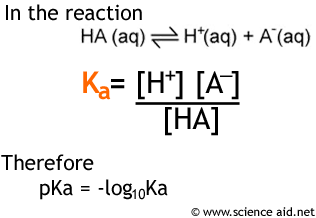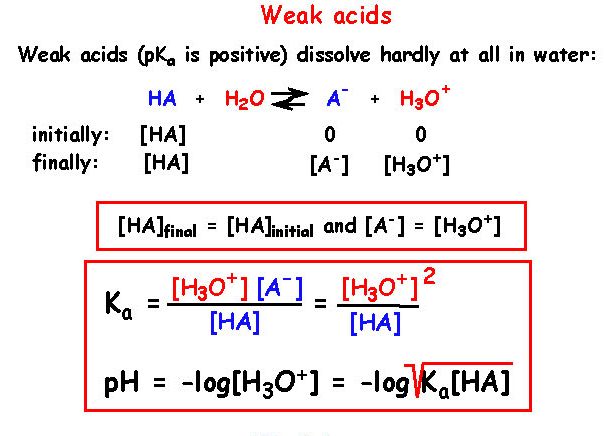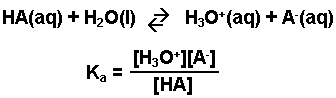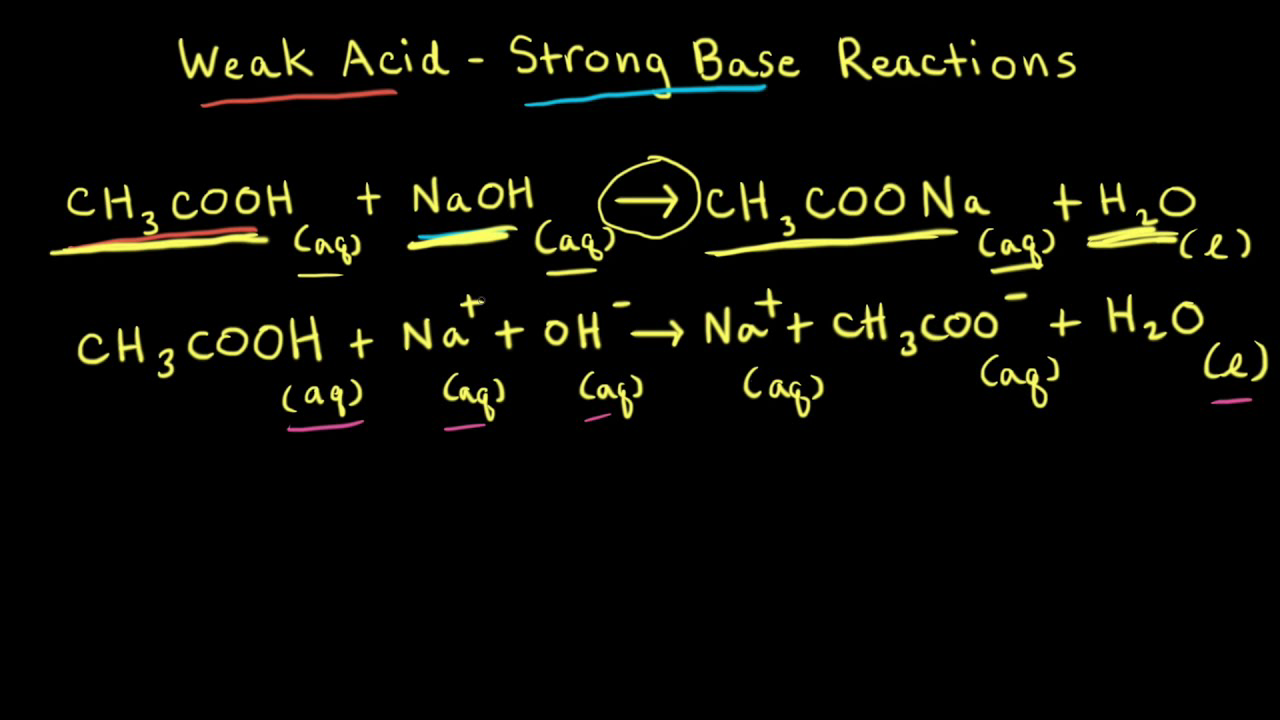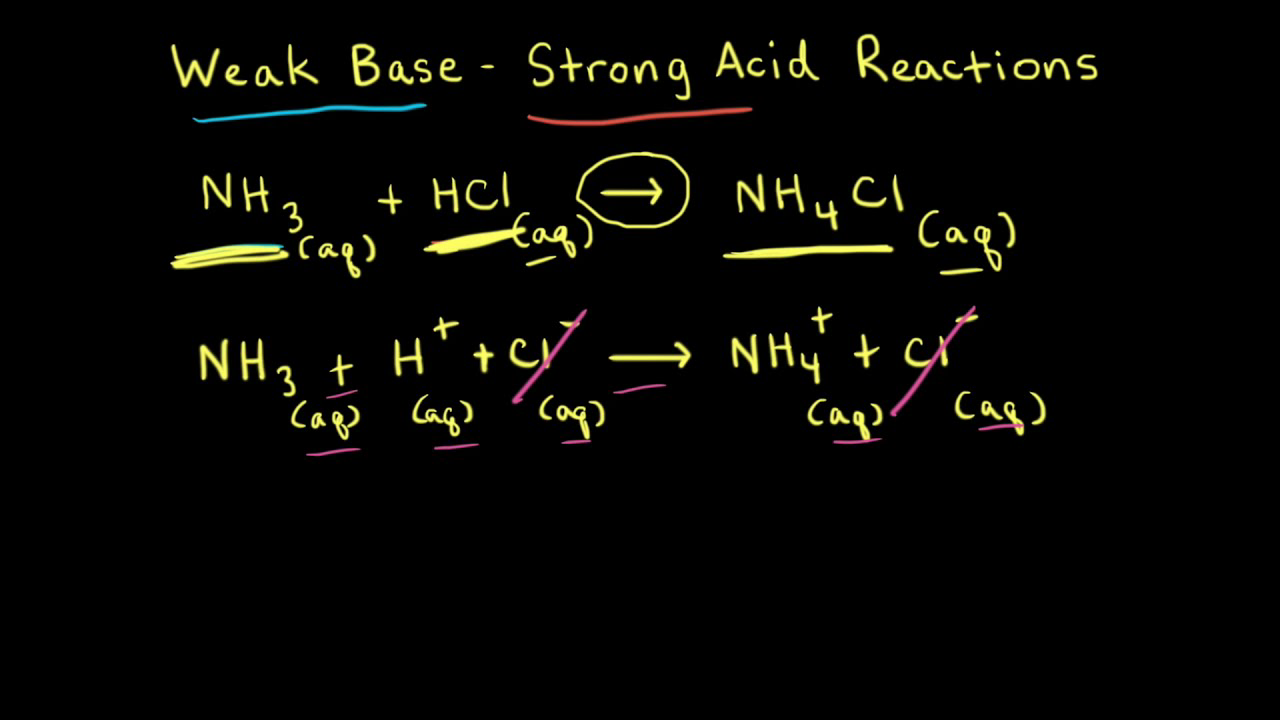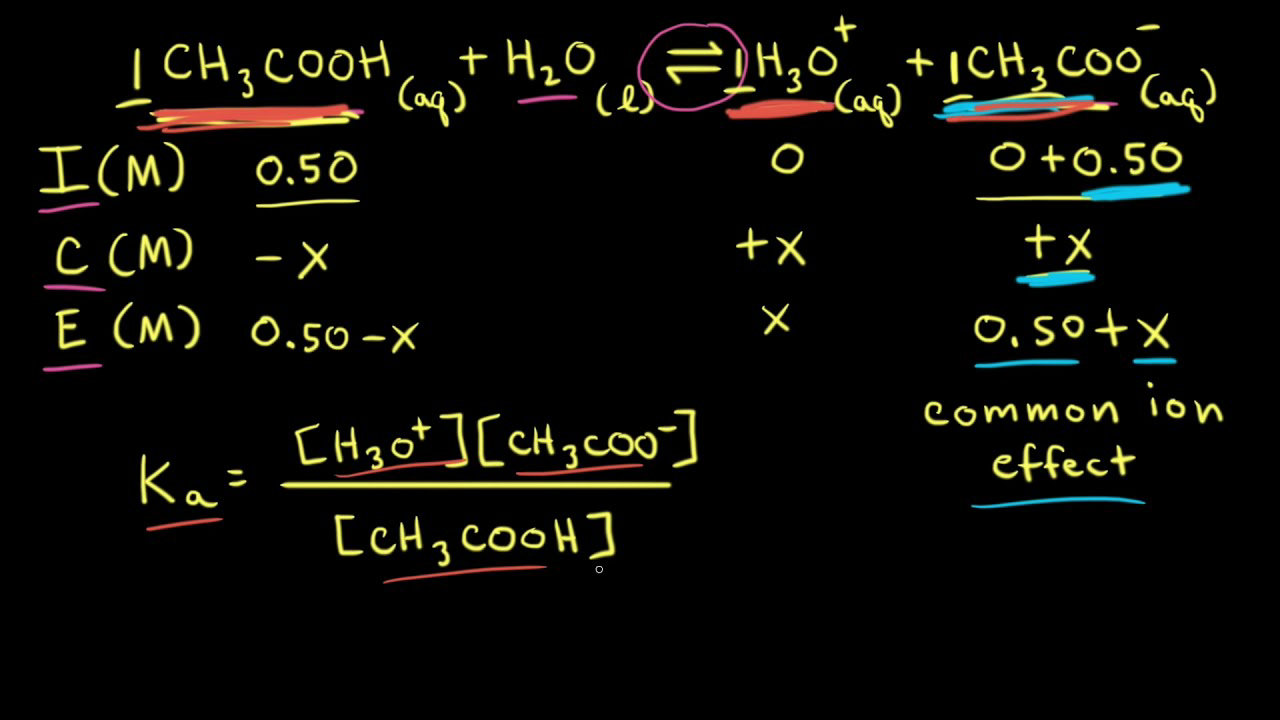
Worked example: Calculating the pH after a weak acid–strong base reaction (excess acid) (video) | Khan Academy
The pH of an acetic acid solution is 3.26. What is the concentration of acetic acid and what is the percent of acid that's ionized? - Quora