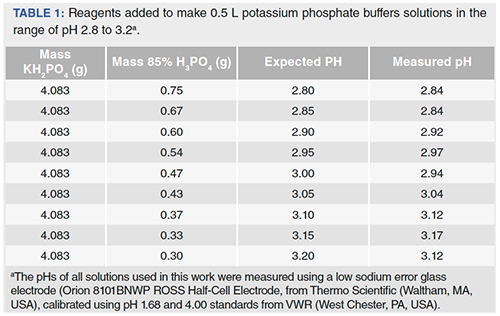
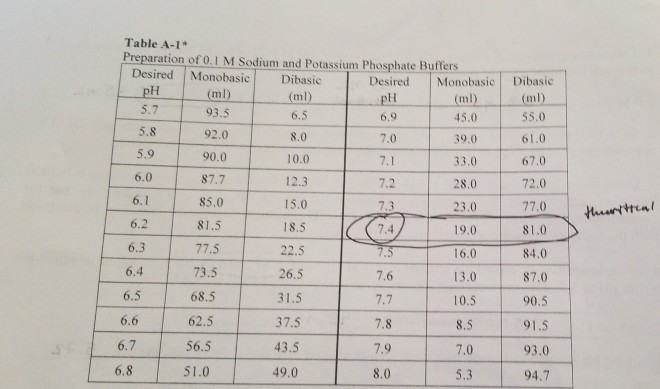
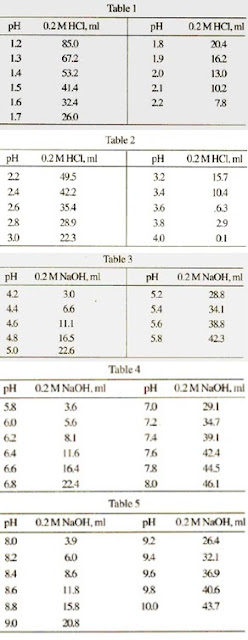
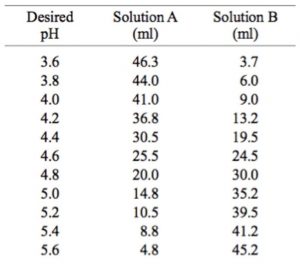
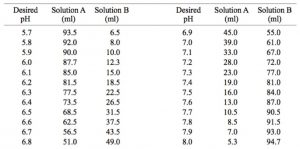
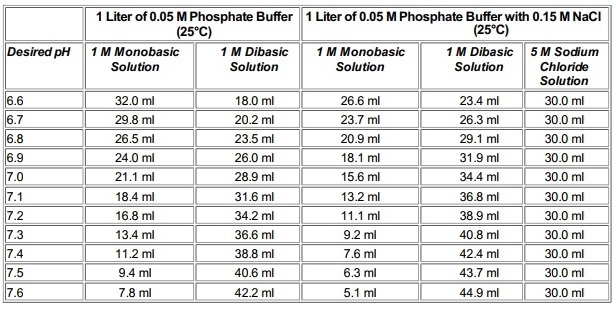
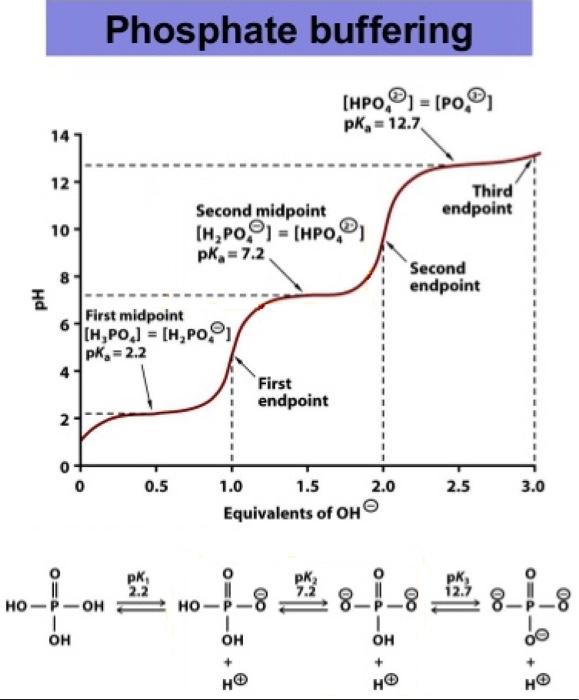
velocities were obtained in potassium phosphate buffer at 30 mC, pH... | Download Scientific Diagram

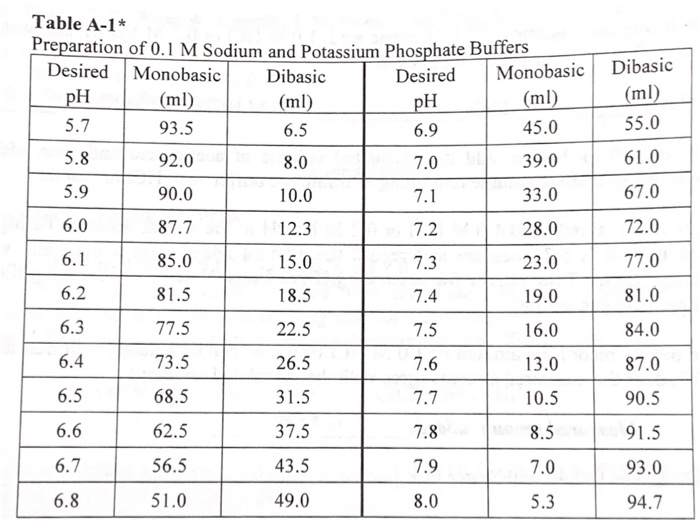
SOLVED: Calculate the amount of monobasic potassium phosphate needed to prepare 100 mL ofa M solution Molecular formula: KHZPOa Molecular Mass: 136.09 glmol Calculate the amount of dibasic potassium phosphate needed to