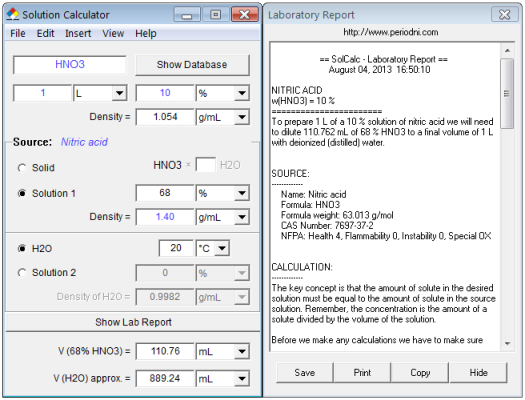
Concentrated `HNO_(3)` is 69% by mass of nitric acid. Calculate the volume of the solution which - YouTube
A certain solution contains 16.9 g of HNO3 dissolved in 125 mL of a substantive solution. Water is added until the volume is 175 mL. What is the molarity of Hno3 in
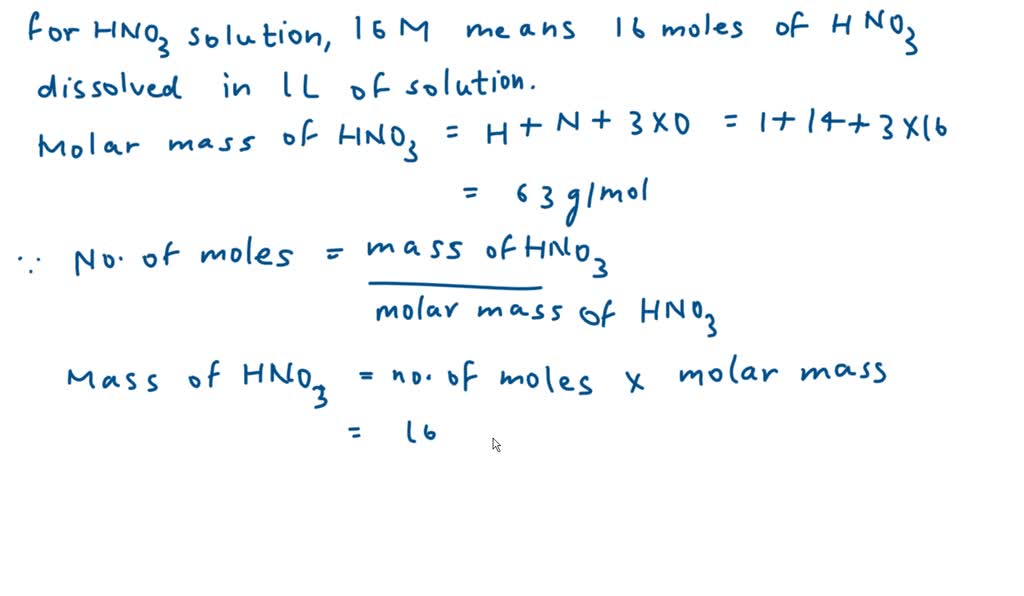
SOLVED: Commercial nitric acid is 16 M HNO3 and it has density of 1.42 g/mL Calculate the masspercent of HNOz in commercial nitric acid?

Question Video: Calculating the Concentration of Nitric Acid via Titrating against a Known Volume of Potassium Hydroxide | Nagwa

SOLVED: Q22: Calculate the molar concentration of HNO3 (63.0 g/mol) in a solution that has a density of 1.42Kg/L and is 70.5% HNO3 (w/w) (Answer ~ 16M)

Calculate the concentration of nitric acid in moles per litre in a sample which has a density - YouTube

Calculate the concentration of nitric acid in moles per litre in a sample which has a density 1.41 g //mL and the mass percent of nitric acid in it being 69%.

Calculate the concentration of nitric acid in moles per litre in a sample which has a density 1.41 g mL^-1 and the mass per cent of nitric acid in it being 69% .





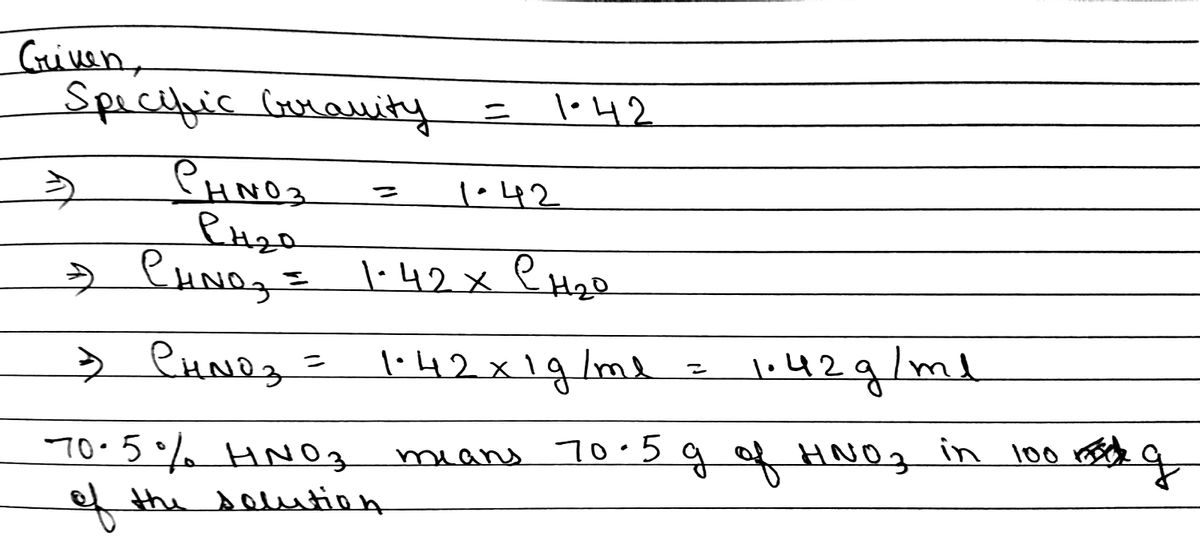


![Solved For a 0.12 M solution of HNO3, calculate: [+], pH, | Chegg.com Solved For a 0.12 M solution of HNO3, calculate: [+], pH, | Chegg.com](https://media.cheggcdn.com/media/6a7/6a7384ad-d27d-4f6c-ba66-edb5228d98b9/php7isMK0.png)