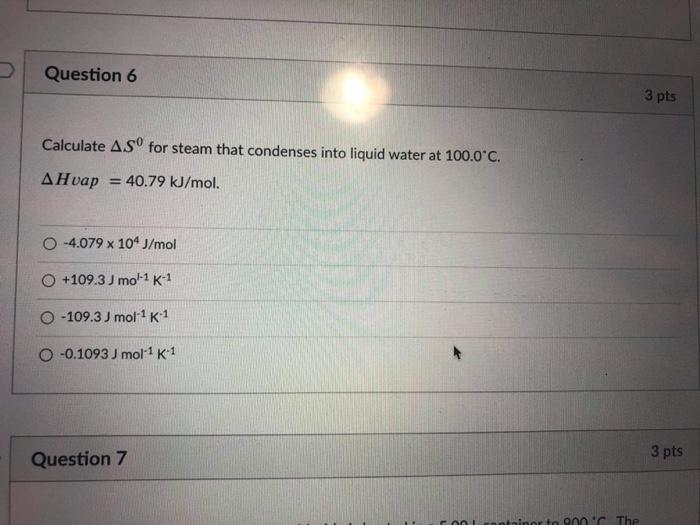
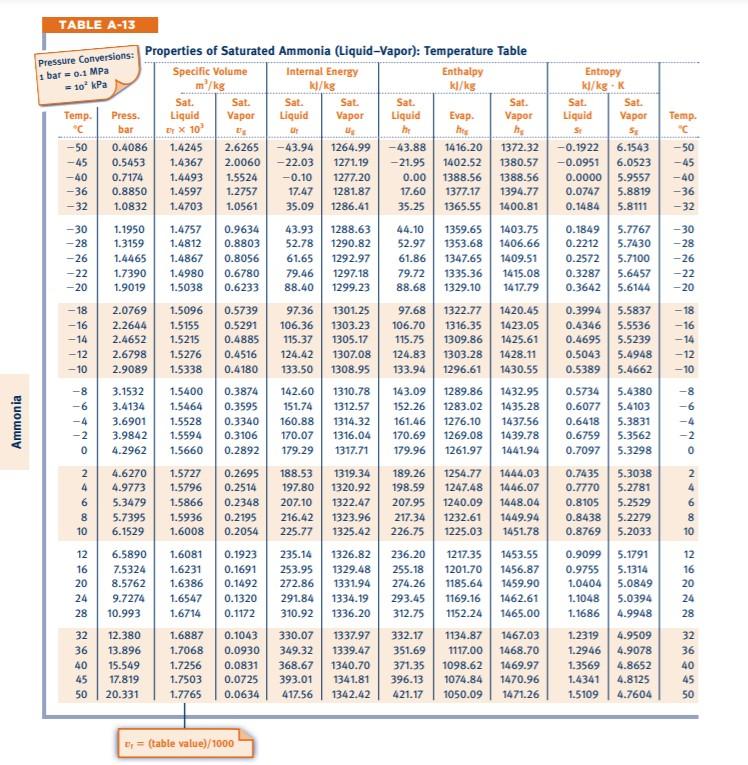
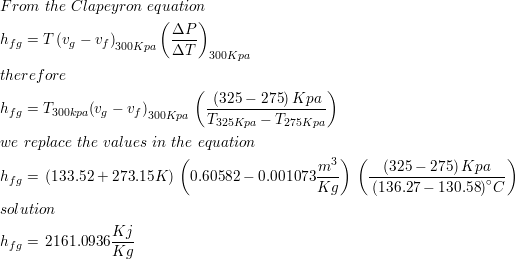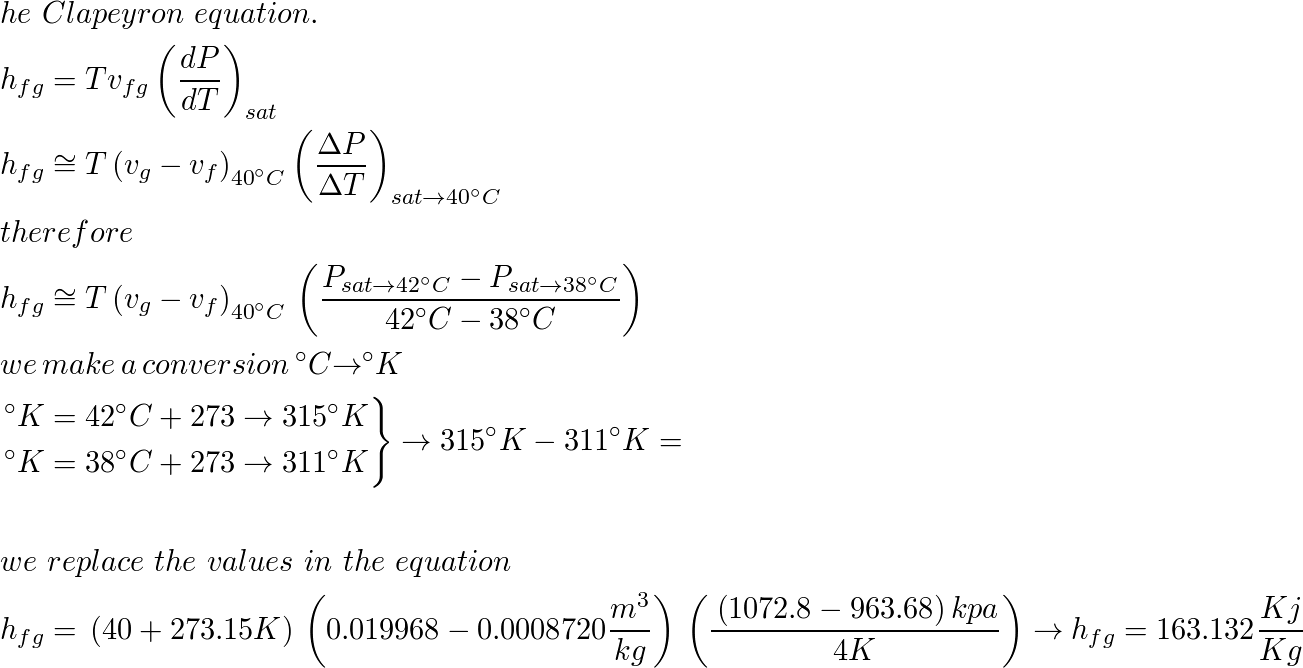Characteristic equations for saturated and superheated steam . and included the specific volumes ofsaturated and superheated steam. To determine the validity of Battellis discussion Knob-lauch, Linde, and Klebe-1- conducted a series of

Knowing the vapor pressure of water is 40.7kJ/mol,calculate the vapor pressure of water at 58 degrees Celsius. | Homework.Study.com

SOLVED: TUTOR Determining Enthalpy of' Vaporization from Terperature and Pressure The Clausius-Clapeyron equation relates the vapor pressure of liquid t0 the ternperalure and the vaporization enthalpy: The equation can be written in
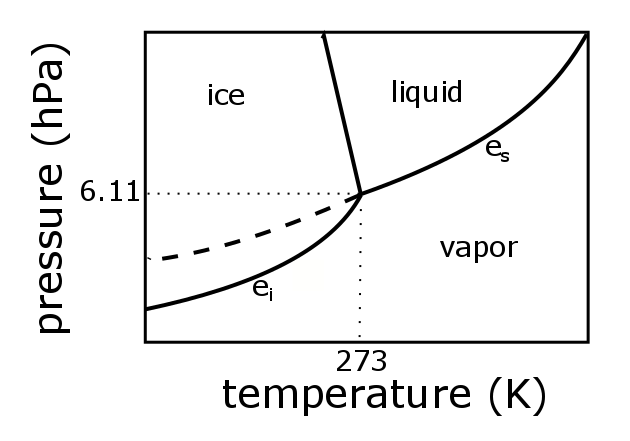
3.3 Phase Diagram for Water Vapor: Clausius Clapeyron Equation | METEO 300: Fundamentals of Atmospheric Science